4.5 - Quantum Physics
Photons
A quantum (plural: quanta) is a small discrete unit of energy.
A photon is a quantum of EM radiation.
Photon energies are always emitted in multiples of the Planck constant, h = 6.63e-34. Photon energy in joules is determined using:
These energies can also be expressed in electronvolts by dividing the energy in joules by e=1.6e-19. One electronvolt is defined as the kinetic energy gained by an electron when it is accelerated through a potential difference of 1V.
The Photoelectric Effect
Each electron can only absorb one photon. This is known as the one-to-one relationship between electrons and photons.
Every metallic surface has a work function phi, which defines the minimum energy required for an electron to overcome electrostatic attraction between it and the metal cations, enabling it to be released/liberated from the metal surface. This energy can be obtained by absorbing a photon:
... where hf is the energy of the photon, phi is the work function, and KEmax is the maximum additional energy from the photon transferred to the photoelectron's kinetic energy store. There is a maximum additional energy, as it is not the case that all photons from the same source have the same energy.
As light frequency is directly proportional to photon energy, the frequency of the EM radiation incident on a metal surface can affect whether or not electrons are liberated. This means a metal surface can also have a threshold frequency.
However, light intensity does not affect electron liberation - it only affects the number of photons released from the radiation source. For radiation above the threshold frequency, however, the rate of emission of photoelectrons is directly proportional to radiation intensity.
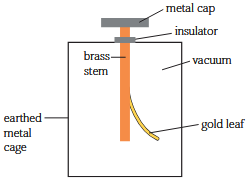
The photoelectric effect can be demonstrated using a gold leaf setup:
- The electroscope is negatively charged, causing the brass stem and gold leaf to be negatively charged, repelling each other, causing the gold leaf to rise.
- EM radiation above the threshold frequency is incident on the metal cap.
- Electrons are liberated from the surface.
- Electrons are transferred from the brass stem and gold leaf to the metal cap.
- The reduction in strength of like negative charge reduces the electrostatic repulsion between the gold leaf and stem.
- The gold leaf falls.
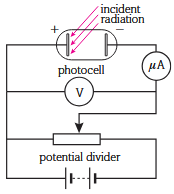
Circuit setup
The stopping potential is the specific negative voltage applied to the collector plate that provides just enough electrical work to cancel out the maximum kinetic energy of the incoming photoelectrons, bringing them to rest exactly at the plate's surface. This can be determined by rearranging the following equation into the form y = mx + c:
|
\raggedright{ \text{We know:}\\
\text{So:}\\
} |
Plotting a graph of stopping potential against radiation frequency can enable one to determine the value of h using the gradient multiplied by e = 1.6e-19.
Wave-Particle Duality
This concept describes the idea that light, other types of EM radiation, and even matter could behave as both a wave and a stream of particles.
This can be observed by de Broglie's experiment involving the firing of electrons at a layer of graphite behind a screen. The result is concentric rings of alternating intensity:
This showed that electrons could diffract and interfere constructively and destructively. However, this was thought to be a behavior exclusive to waves.
The (de Broglie) wavelength of a particle can be determined with:
You can also substitute:
Wavelength is inversely proportional to mass. Since a wavelength similar to aperture size is required for diffraction, you wouldn't see a human diffracting walking through a door, for example. Assuming a mass 75kg and velocity 1ms-1:
... which is nowhere close to the width of any human's body, let alone the width of a doorway.
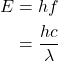
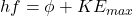
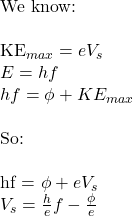

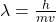
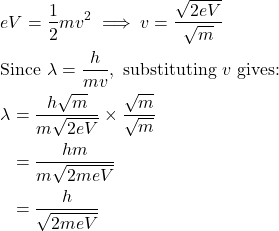
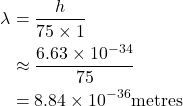
No comments to display
No comments to display