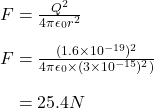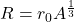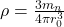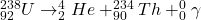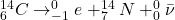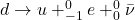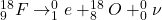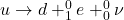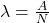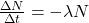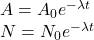6.4 - Nuclear and Particle Physics
The Nuclear Atom
The alpha particle scattering experiment involved the firing of alpha particles at a thin sheet of gold foil. Due to the old belief in the plum-pudding model for the atom, it was expected that the alpha particles would pass straight through the sheet due to their momentum.
Actual observations:
- Most passed through.
- Some were deflected at an angle.
- Some reflected completely back towards the detector's direction.
Deductions:
- The atom is mostly empty space.
- The vast majority of the atom's mass is concentrated at the centre of the atom (the nucleus).
- There is a very small, concentrated point of positive charge at the centre of the atom (the nucleus).
Bohr's "nuclear model" of the atom involves electrons orbiting certain orbits/energy levels around a central nucleus (that contains the vast majority of the mass).
Isotopes are atoms of an element with the same number of protons but a different number of neutrons. The term nuclide refers to a specific species of atom. E.g. C-12 and C-14 are different isotopes of carbon, but a C-12 nuclide has two less neutrons than a C-14 nuclide.
The Strong Nuclear Force
Using Coulomb's law, we can calculate the electric force between two protons at 3fm of separation:
This is an extremely large force for protons this close to each other. The gravitational force is far too small considering how low the mass of a proton is. The actual force counteracting this electric force is known as the strong nuclear force.
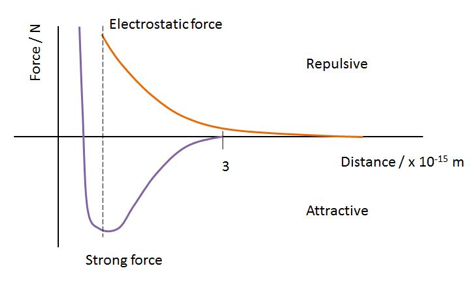
This force acts within the confines of the nucleus, but decreases rapidly with distance and does not extend much beyond adjacent protons and neutrons. The force must act between nucleons, and is independent of charge. At a particular distance, however, it turns from an attractive force to a repulsive force, otherwise the nucleus would collapse on itself:
The strong nuclear force, as you can see, stops acting just a little before 4fm of nucleon separation.
Nuclear Density
As the number of nucleons increases, the radius of an atom increases at a decreasing rate, reaching a constant-looking positive gradient on a radius x nucleon graph. The radius of a nucleus can be determined using:
... where r0 is a constant and A is the nucleon number of the atom. r0 is approximately 1.05fm = 1.05e-15m.
Nuclear density is equal to:
... where mn is the mass of a nucleon. Since all of these values are constants, all nuclei have the same density.
Fundamental Particles
- Hadrons are particles consisting of a combination of quarks to give a net zero or whole number charge, and can be subject to the strong nuclear force. E.g. neutrons and protons.
- Leptons are a type of fundamental particle. E.g. electrons and neutrinos. Each charged lepton has its own neutrino version, e.g. an electron neutrino or a muon neutrino.
- Quarks are fundamental particles that are components of hadrons. E.g. up and down quarks.
- The weak nuclear force is experienced by both quarks and leptons, and is responsible for beta decay due to the ability to change quarks' types and leptons' types.
- An antiparticle is a particle of antimatter that has the same rest mass as the original with an equal and opposite charge. When they collide with their original, they annihilate, turning into energy as per E = mc2 as two gamma photons in opposite directions.
The three quarks that were initially proposed were called the "up", "down", and "strange" quarks:
| Quarks | Antiquarks | |||||
| Type | up | down | strange | anti-up | anti-down | anti-strange |
| Symbol | u | d | s | u bar | d bar | s bar |
| Charge Q | +2e/3 | -1e/3 | -1e/3 | -2e/3 | +1e/3 | +1e/3 |
| Strangeness S | 0 | 0 | -1 | 0 | 0 | +1 |
| Baryon number B | 1/3 | 1/3 | 1/3 | -1/3 | -1/3 | -1/3 |
We can use these quarks to see how certain particles and antiparticles are constructed:
| Particle | Antiparticle |
| Proton: uud | Anti-Proton: u-bar d-bar d-bar |
| Neutron: udd | Anti-Neutron: u-bar u-bar d-bar |
When a proton decays to a neutron, it just means an up quark of a proton decays to a down quark. Since the difference in charge of an up and down quark is -1e, an electron is released - beta minus decay.
Radioactivity
Radioactive decay is the spontaneous and random decay of an unstable nucleus by the emission of alpha, beta, and gamma radiation. The word "spontaneous" is used because decay is not affected by any external factors, such as temperature, pressure, chemical reactions, and magnetic fields. Neither the exact number of decays per second nor the fact that a particle will decay can be estimated.
| Type | Description |
| Alpha |
Alpha decay example: |
| Beta |
Beta minus decay example and quark equation: Beta plus decay example and quark equation:
|
| Gamma |
|
Radioactive Decay Equations and Half Life
The activity dictates the number of nuclear decays per unit time. One decay per second is known as 1Bq (becquerel).
The decay constant lambda is the probability that an individual nucleus will decay per unit time.
... where A is activity and N is the number of undecayed nuclei. The rate of change of undecayed nuclei is:
General decay equations for exponential decrease from initial values A0 and N0 are:
You can rearrange these using ln and log laws in order to find lambda using graphical methods.
A substance's half life is the amount of time taken for the activity of a substance or the number of undecayed nuclei to halve. Can be estimated with a Nxt graph. You can substitute A = 0.5A0 into the above exponential equation to find the half life given the value of lambda.
Radioactive Dating
Carbon Dating
Organisms take in carbon dioxide from the atmosphere. A small fraction of carbon atoms in this CO2 is the radioactive C-14 isotope of carbon rather than the normal C-12 isotope. Once an organism dies, it no longer takes in CO2, so the C-14 starts to decay into nitrogen.
The ratio of C-14 to C-12 decreases over time, and can be compared with the ration of a currently living organism to estimate the age of the organism/object.
However, since the amount of C-14 is so small, count rates are also very small, and after a few half lives may be indistinguishable from the background count rate. It also assumes the ratio of C-14 to C-12 in the atmosphere has always been constant, which may not be true.
Dating Rocks
All rocks contain tiny amounts of radioactive isotopes such as U-238 and Ru-87, which have very long half lives. Relative proportions of the parent atoms and decay products can be used to estimate age.
Generating Energy
Nuclear Fission
Nuclear fission is when a radioactive nucleus absorbs a high-energy neutron, causing it to split into two more-stable nuclei and release 2-3 high-energy neutrons and gamma radiation. (Extra: Energy also comes from KE of daughter nuclei, which collide with moderator particles to transfer energy.)
e.g. uranium 235 (insert example here)
A nuclear fission reactor consists of a solid concrete wall, a moderator, and a reaction chamber in the moderator. Inside the reaction chamber, there are numerous control rods and pills containing the radioactive substance.
(diagram here)
- The concrete wall ensures that radiation cannot escape the reaction chamber, keeping employees safe from radiation exposure.
- The moderator reduces the speed of high-energy neutrons so that they can be absorbed by nuclei. (There was another advantage)
- The control rods are made of a material such as boron, and exist to absorb excess high-energy neutrons. This is to keep control of the chain reaction. Losing control leads to reactor meltdowns such as Chernobyl.
Nuclear Fusion
Nuclear fusion is when two nuclei are fused together, releasing energy