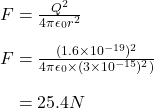6.4 - Nuclear and Particle Physics
The Nuclear Atom
The alpha particle scattering experiment involved the firing of alpha particles at a thin sheet of gold foil. Due to the old belief in the plum-pudding model for the atom, it was expected that the alpha particles would pass straight through the sheet due to their momentum.
Actual observations:
- Most passed through.
- Some were deflected at an angle.
- Some reflected completely back towards the detector's direction.
Deductions:
- The atom is mostly empty space.
- The vast majority of the atom's mass is concentrated at the centre of the atom (the nucleus).
- There is a very small, concentrated point of positive charge at the centre of the atom (the nucleus).
Bohr's "nuclear model" of the atom involves electrons orbiting certain orbits/energy levels around a central nucleus (that contains the vast majority of the mass).
Isotopes are atoms of an element with the same number of protons but a different number of neutrons. The term nuclide refers to a specific species of atom. E.g. C-12 and C-14 are different isotopes of carbon, but a C-12 nuclide has two less neutrons than a C-14 nuclide.
The Strong Nuclear Force
Using Coulomb's law, we can calculate the electric force between two protons at 3fm of separation:
This is an extremely large force for protons this close to each other. The gravitational force is far too small considering how low the mass of a proton is. The actual force counteracting this electric force is known as the strong nuclear force.
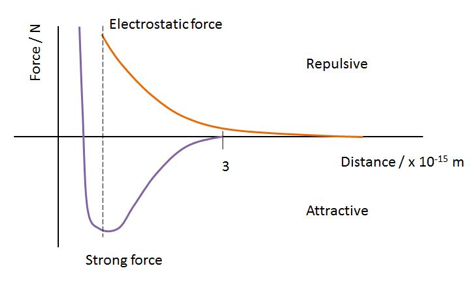
This force acts within the confines of the nucleus, but decreases rapidly with distance and does not extend much beyond adjacent protons and neutrons. The force must act between nucleons, and is independent of charge. At a particular distance, however, it turns from an attractive force to a repulsive force, otherwise the nucleus would collapse on itself:
The strong nuclear force, as you can see, stops acting just a little before 4fm of nucleon separation.